Sirnormanfosterene is a gherkin-shaped arrangement of 928 carbon atoms found naturally in soot.
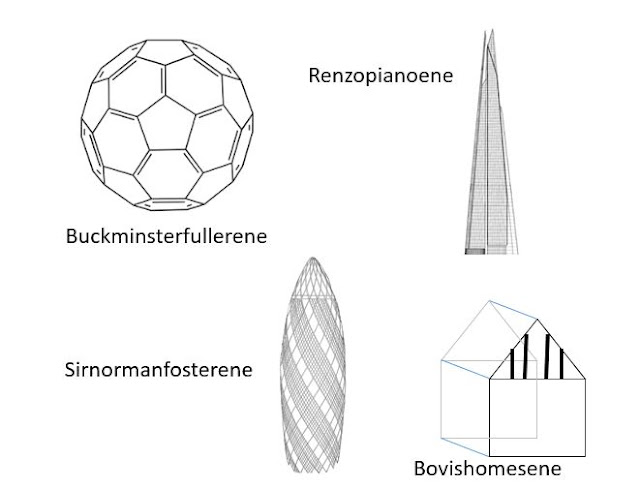 Renzopianoene is a tall molecule with a tapering square cross-section containing 2048 atoms of carbon. It does not occur naturally on Earth, as far as we know, and was created in my laboratories using laser deposition interferometry techniques.
Renzopianoene is a tall molecule with a tapering square cross-section containing 2048 atoms of carbon. It does not occur naturally on Earth, as far as we know, and was created in my laboratories using laser deposition interferometry techniques.However, the most remarkable carbon molecule we have encountered is bovishomesene, a collection of fourteen atoms forming a cube surmounted by a triangular prism. Particularly strong co-valent bonds between p-shell electrons in the atoms at the 'gable' of the prism creates a stunning mock-Tudor effect. When Bovishomesene molecules are amassed in significant quantities they assume a pseudo-crystalline formation known as an 'e-state', which mimics the principles of Penrose tiling with an extremely high packing density. Bovishomesene molecules- or 'bovvy boxes' as they are known in my labs- decay naturally over a period of ten to twenty years.
Such a great post to know different useful information https://oneworldrental.ae/event-wi-fi-rental
ReplyDelete